Search
- Page Path
- HOME > Search
Original Articles
- Calcium & Bone Metabolism
- Real-World Safety and Effectiveness of Denosumab in Patients with Osteoporosis: A Prospective, Observational Study in South Korea
- Yumie Rhee, Dong-Gune Chang, Jeonghoon Ha, Sooa Kim, Yusun Lee, Euna Jo, Jung-Min Koh
- Endocrinol Metab. 2022;37(3):497-505. Published online June 3, 2022
- DOI: https://doi.org/10.3803/EnM.2022.1427
- 5,404 View
- 268 Download
- 8 Web of Science
- 8 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Background
The efficacy and safety of denosumab have been established in a phase 3, randomized, placebo-controlled trial in Korean postmenopausal women with osteoporosis. This postmarketing surveillance study was aimed to investigate the safety and effectiveness of denosumab in Korean real-world clinical practice.
Methods
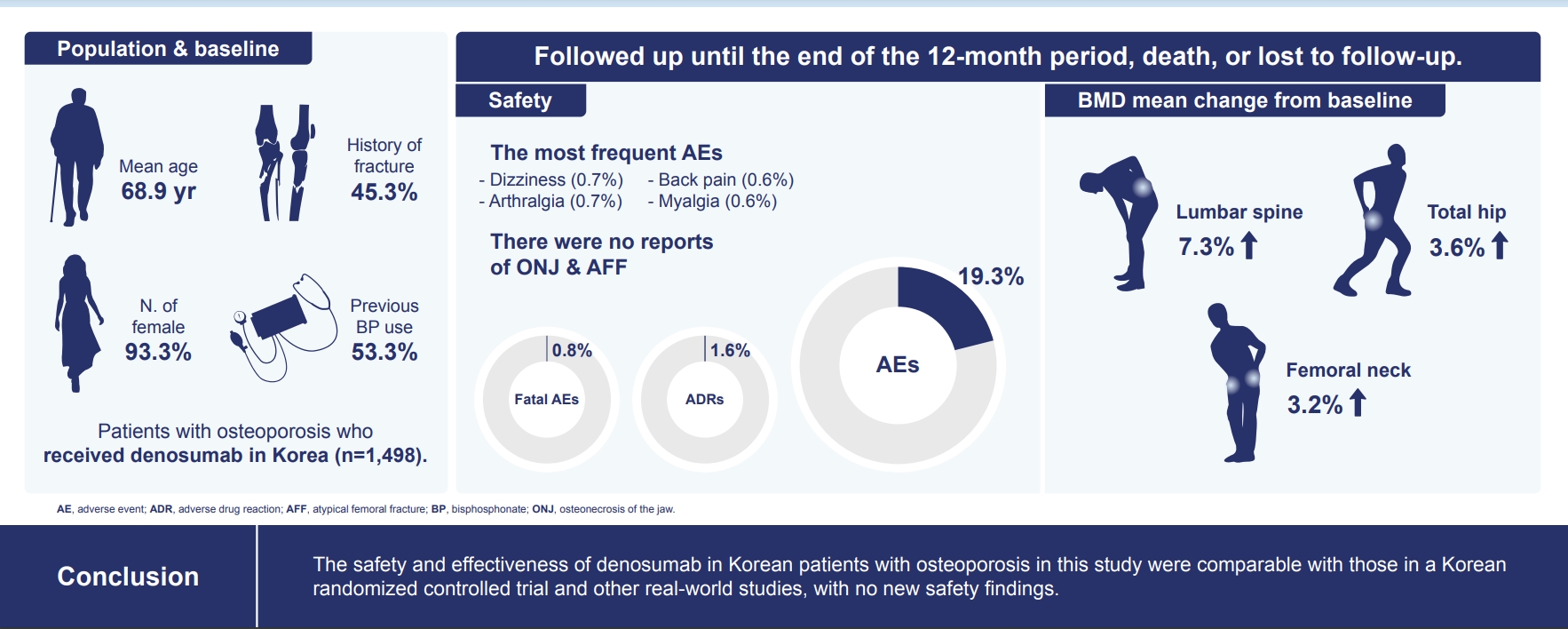
Patients with osteoporosis who had received denosumab per the Korean approved indications in the postmarketing setting between September 2014 and September 2019 were enrolled. The primary endpoint was the incidence of adverse events (AEs) and adverse drug reactions (ADRs). The secondary endpoint was the percent change from baseline in bone mineral density (BMD) of the lumbar spine, total hip, and femoral neck.
Results
Of the 3,221 patients enrolled, 3,185 were included in the safety analysis set; 2,973 (93.3%) were female, and the mean± standard deviation (SD) age was 68.9±9.9 years. The mean±SD study period was 350.0±71.4 days. AEs, fatal AEs, and ADRs occurred in 19.3%, 0.8%, and 1.6%, respectively. The most frequent AEs, occurring in >0.5% of patients, were dizziness (0.7%), arthralgia (0.7%), back pain (0.6%), and myalgia (0.6%). Hypocalcemia occurred in 0.3% of patients. There were no cases of osteonecrosis of the jaw and atypical femoral fracture. Mean±SD percent change from baseline in BMD of the lumbar spine, total hip, and femoral neck was 7.3%±23.6%, 3.6%±31.4%, and 3.2%±10.7%, respectively.
Conclusion
The safety and effectiveness of denosumab in Korean patients with osteoporosis in this study were comparable with those in the Korean randomized controlled trial, with no new safety findings. -
Citations
Citations to this article as recorded by- Prevalence of denosumab-induced hypocalcemia: a retrospective observational study of patients routinely monitored with ionized calcium post-injection
Anna Spångeus, Johan Rydetun, Mischa Woisetschläger
Osteoporosis International.2024; 35(1): 173. CrossRef - Cost-consequence analysis of continuous denosumab therapy for osteoporosis treatment in South Korea
Seungju Cha, Minjeong Sohn, Hyowon Yang, Eric J. Yeh, Ki-Hyun Baek, Jeonghoon Ha, Hyemin Ku
BMC Musculoskeletal Disorders.2024;[Epub] CrossRef - Denosumab and the Risk of Diabetes in Patients Treated for Osteoporosis
Huei-Kai Huang, Albert Tzu-Ming Chuang, Tzu-Chi Liao, Shih-Chieh Shao, Peter Pin-Sung Liu, Yu-Kang Tu, Edward Chia-Cheng Lai
JAMA Network Open.2024; 7(2): e2354734. CrossRef - Adverse Effects of Denosumab in Kidney Transplant Recipients: A 20-Year Retrospective Single-Center Observation Study in Central Taiwan
Tsung-Yin Tsai, Zi-Hong You, Shang-Feng Tsai, Ming-Ju Wu, Tung-Min Yu, Ya-Wen Chuang, Yung-Chieh Lin, Ya-Lian Deng, Chiann-Yi Hsu, Cheng-Hsu Chen
Transplantation Proceedings.2023; 55(4): 837. CrossRef - Persistence with Denosumab in Male Osteoporosis Patients: A Real-World, Non-Interventional Multicenter Study
Chaiho Jeong, Jeongmin Lee, Jinyoung Kim, Jeonghoon Ha, Kwanhoon Jo, Yejee Lim, Mee Kyoung Kim, Hyuk-Sang Kwon, Tae-Seo Sohn, Ki-Ho Song, Moo Il Kang, Ki-Hyun Baek
Endocrinology and Metabolism.2023; 38(2): 260. CrossRef - Effect of Denosumab on Bone Density in Postmenopausal Osteoporosis: A Comparison with and without Calcium Supplementation in Patients on Standard Diets in Korea
Chaiho Jeong, Jinyoung Kim, Jeongmin Lee, Yejee Lim, Dong-Jun Lim, Ki-Hyun Baek, Jeonghoon Ha
Journal of Clinical Medicine.2023; 12(21): 6904. CrossRef - Denosumab
Reactions Weekly.2022; 1919(1): 221. CrossRef - Denosumab, an effective osteoporosis treatment option for men
Sung Hye Kong
The Korean Journal of Internal Medicine.2022; 37(5): 947. CrossRef
- Prevalence of denosumab-induced hypocalcemia: a retrospective observational study of patients routinely monitored with ionized calcium post-injection

- Clinical Study
- Romosozumab in Postmenopausal Korean Women with Osteoporosis: A Randomized, Double-Blind, Placebo-Controlled Efficacy and Safety Study
- Ki-Hyun Baek, Yoon-Sok Chung, Jung-Min Koh, In Joo Kim, Kyoung Min Kim, Yong-Ki Min, Ki Deok Park, Rajani Dinavahi, Judy Maddox, Wenjing Yang, Sooa Kim, Sang Jin Lee, Hyungjin Cho, Sung-Kil Lim
- Endocrinol Metab. 2021;36(1):60-69. Published online February 24, 2021
- DOI: https://doi.org/10.3803/EnM.2020.848
- 6,804 View
- 390 Download
- 7 Web of Science
- 10 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
This phase 3 study evaluated the efficacy and safety of 6-month treatment with romosozumab in Korean postmenopausal women with osteoporosis.
Methods
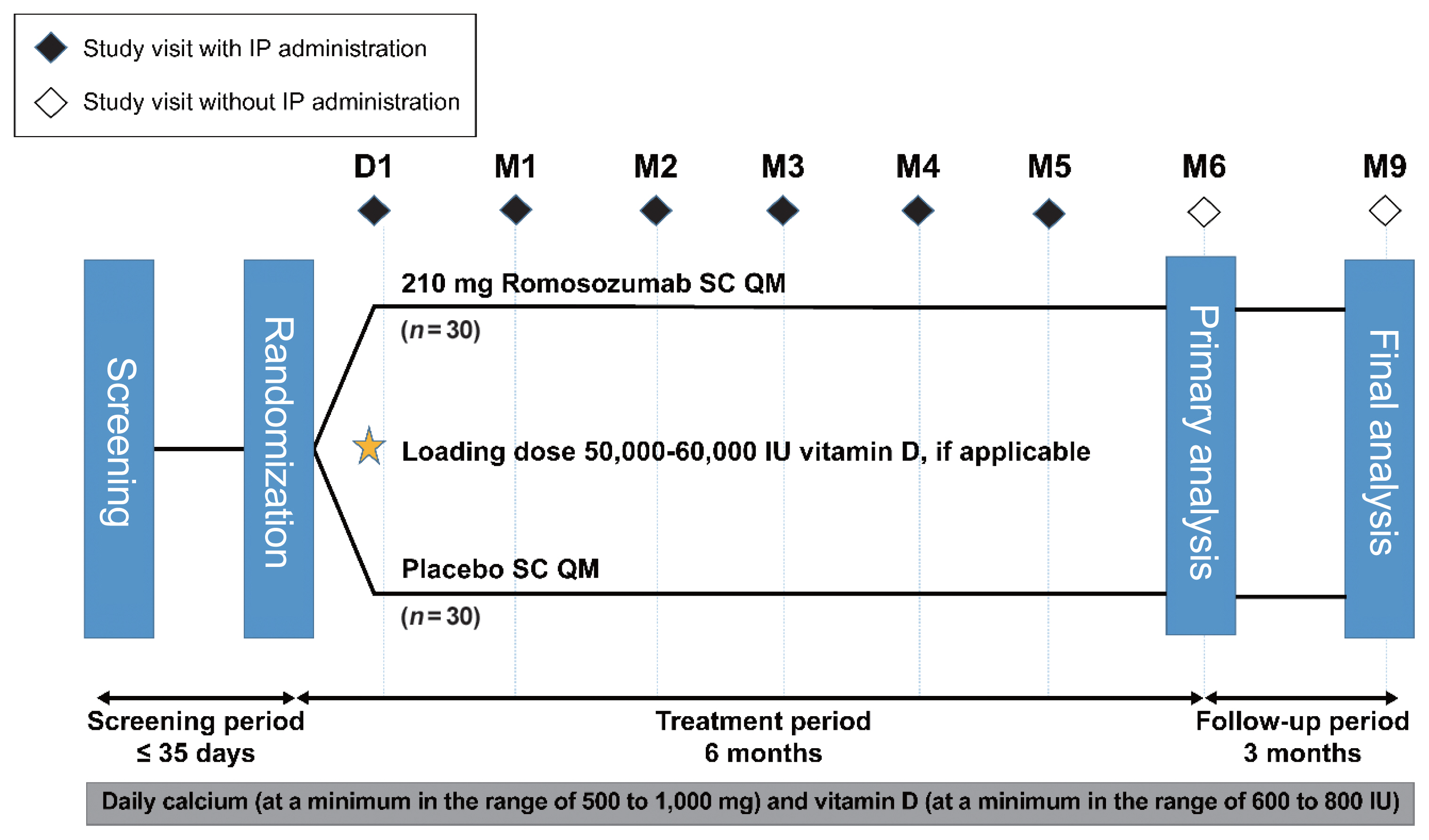
Sixty-seven postmenopausal women with osteoporosis (bone mineral density [BMD] T-scores ≤–2.5 at the lumbar spine, total hip, or femoral neck) were randomized (1:1) to receive monthly subcutaneous injections of romosozumab (210 mg; n=34) or placebo (n=33) for 6 months.
Results
At month 6, the difference in the least square (LS) mean percent change from baseline in lumbar spine BMD (primary efficacy endpoint) between the romosozumab (9.5%) and placebo (–0.1%) groups was significant (9.6%; 95% confidence interval, 7.6 to 11.5; P<0.001). The difference in the LS mean percent change from baseline was also significant for total hip and femoral neck BMD (secondary efficacy endpoints). After treatment with romosozumab, the percent change from baseline in procollagen type 1 N-terminal propeptide transiently increased at months 1 and 3, while that in C-terminal telopeptide of type 1 collagen showed a sustained decrease. No events of cancer, hypocalcemia, injection site reaction, positively adjudicated atypical femoral fracture or osteonecrosis of the jaw, or positively adjudicated serious cardiovascular adverse events were observed. At month 9, 17.6% and 2.9% of patients in the romosozumab group developed binding and neutralizing antibodies, respectively.
Conclusion
Treatment with romosozumab for 6 months was well tolerated and significantly increased lumbar spine, total hip, and femoral neck BMD compared with placebo in Korean postmenopausal women with osteoporosis (ClinicalTrials.gov identifier NCT02791516). -
Citations
Citations to this article as recorded by- A pharmacovigilance analysis of FDA adverse event reporting system events for romosozumab
Zepeng Chen, Ming Li, Shuzhen Li, Yuxi Li, Junyan Wu, Kaifeng Qiu, Xiaoxia Yu, Lin Huang, Guanghui Chen
Expert Opinion on Drug Safety.2023; 22(4): 339. CrossRef - Evaluation of the efficacy and safety of romosozumab (evenity) for the treatment of osteoporotic vertebral compression fracture in postmenopausal women: A systematic review and meta‐analysis of randomized controlled trials (CDM‐J)
Wenbo Huang, Masashi Nagao, Naohiro Yonemoto, Sen Guo, Takeshi Tanigawa, Yuji Nishizaki
Pharmacoepidemiology and Drug Safety.2023; 32(6): 671. CrossRef - Efficacy and Cardiovascular Safety of Romosozumab: A Meta-analysis and Systematic Review
Seo-Yong Choi, Jeong-Min Kim, Sang-Hyeon Oh, Seunghyun Cheon, Jee-Eun Chung
Korean Journal of Clinical Pharmacy.2023; 33(2): 128. CrossRef - Clinical Studies On Romosozumab: An Alternative For Individuals With A High Risk Of Osteoporotic Fractures: A Current Concepts Review (Part I)
E. Carlos Rodriguez-Merchan, Alonso Moreno-Garcia, Hortensia De la Corte-Rodriguez
SurgiColl.2023;[Epub] CrossRef - Romosozumab in osteoporosis: yesterday, today and tomorrow
Dong Wu, Lei Li, Zhun Wen, Guangbin Wang
Journal of Translational Medicine.2023;[Epub] CrossRef - Efficacy and safety of anti-sclerostin antibodies in the treatment of osteoporosis: A meta-analysis and systematic review
Frideriki Poutoglidou, Efthimios Samoladas, Nikolaos Raikos, Dimitrios Kouvelas
Journal of Clinical Densitometry.2022; 25(3): 401. CrossRef - Benefits of lumican on human bone health: clinical evidence using bone marrow aspirates
Yun Sun Lee, So Jeong Park, Jin Young Lee, Eunah Choi, Beom-Jun Kim
The Korean Journal of Internal Medicine.2022; 37(4): 821. CrossRef - What is the risk of cardiovascular events in osteoporotic patients treated with romosozumab?
I. R. Reid
Expert Opinion on Drug Safety.2022; 21(12): 1441. CrossRef - Proxied Therapeutic Inhibition on Wnt Signaling Antagonists and Risk of Cardiovascular Diseases: Multi-Omics Analyses
Yu Qian, Cheng-Da Yuan, Saber Khederzadeh, Ming-Yu Han, Hai-Xia Liu, Mo-Chang Qiu, Jian-Hua Gao, Wei-Lin Wang, Yun-Piao Hou, Guo-Bo Chen, Ke-Qi Liu, Lin Xu, David Karasik, Shu-Yang Xie, Hou-Feng Zheng
SSRN Electronic Journal .2022;[Epub] CrossRef - Multi-Omics Analyses Identify Pleiotropy and Causality Between Circulating Sclerostin and Atrial Fibrillation
Yu Qian, Peng-Lin Guan, Saber Khederzadeh, Ke-Qi Liu, Cheng-Da Yuan, Ming-Yu Han, Hai-Xia Liu, Mo-Chang Qiu, Jian-Hua Gao, Wei-Lin Wang, Yun-Piao Hou, Guo-Bo Chen, Lin Xu, David Karasik, Shu-Yang Xie, sheng zhifeng, Hou-Feng Zheng
SSRN Electronic Journal .2022;[Epub] CrossRef
- A pharmacovigilance analysis of FDA adverse event reporting system events for romosozumab


 KES
KES

 First
First Prev
Prev



